Prove compliance and secure international market access
The regulations on the international markets are as comprehensive as the range of different medical devices. In the future, product testing will also be an important and mandatory part to fulfill the MDR requirements. Therefore, familiarize with the applicable legal requirements in your target market at an early stage in order to avoid delays in approval.
Find out on this website how you can prove the safety of your medical devices and thus secure access to international markets. We support you in this process with our tests and certifications for medical devices.
Our Testings for the Security of your Medical Device
Patients and users rely on the safety of your medical devices, so conformity with the applicable requirements of the target market must be clearly ensured.
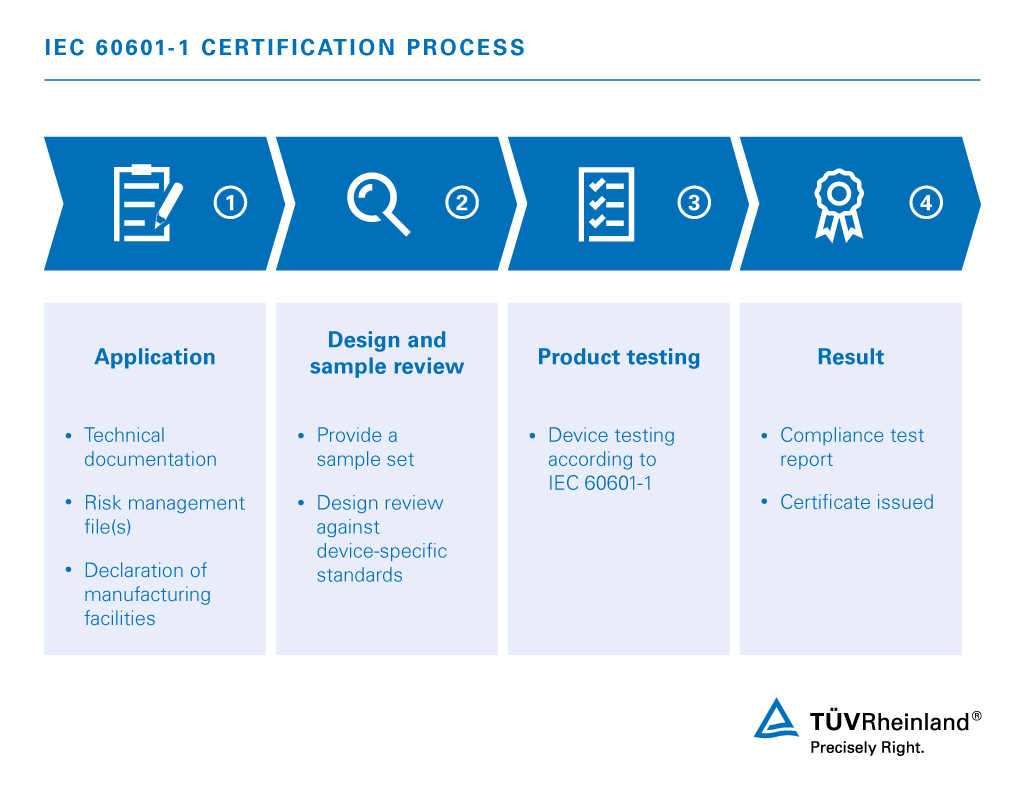
For medical electrical equipment, the IEC 60601-1 series of standards is of particular importance. This harmonized and globally recognized series of standards identifies collateral, particular and performance standards for each type of device. However, there are some local deviations, which significantly increase the complexity for internationally operating manufacturers and retailers.
We are experienced experts in dealing with the IEC 60601-1 series of standards as well as the supplementary IEC/EN 80601 and DIN EN 61010 standards. Thanks to our efficient processes, we are able to perform all required tests quickly and easily. Learn more about testing according to the IEC 60601-1 series of standards in our fact sheet, on our service page or in personal contact with our experts.
| EMC | We test your medical device for electromagnetic compatibility. |
| Batteries | We test and certify your battery according to IEC 60086-1, IEC 60086-2 and national rules and regulations. |
| Wireless | We test and certify IoT medical technology and perform pentesting upon request to ensure cybersecurity. |
| Chemical testing | With chemical testing, you increase confidence in your product. |
| Other (partial) tests for medical devices | We offer mechanical testing, environmental simulation, usability and ergonomics testing, and more. |
| EN ISO 13485 certification | The international standard EN ISO 13485 is the standard for quality management systems in the medical device sector. We review your quality management in areas such as design/development, production, customer service and assembly of medical devices. |
| ISO 15378 certification | We review and certify your quality management system for the production of primary packaging for medicinal and pharmaceutical products based on the applicable standard ISO 15378. |
| ISO 9001, ISO 14001 and ISO 27001 | We certify your quality management system, environmental management or information security. |
| Assessment and certification of companion diagnostics | We support pharmaceutical companies and CDx manufacturers with meeting the requirements of the new IVDR. |
International Approvals of Medical Devices
The world has become smaller in many respects due to globalization. However, many national laws and individual regulations still apply when selling medical devices on international markets.
We support you worldwide with the approval of your medical device. Decide for comprehensive service from a single source - with local contacts who are very familiar with the approval conditions of the respective country. Take advantage of our experience on your way to new markets. Our experts will be happy to inform you which approval procedure is right for your product.
| Market | Our Service | IEC 60601-1 adopted as: |
|---|---|---|
| Brazil | ANVISA Product registration; INMETRO | ABNT NBR IEC 60601-1:2010 + Emenda IEC:2012 |
| China | Testing services regarding NMPA requirements | - |
| Japan | Inspections and audits according to PMD law | JIS T0601-1:2017 |
| Canada | cTUVus mark | CAN/CSA C22.2 No. 60601-1:14 |
| Malaysia | Asia Medical Device Directive (AMDD); Registered conformity assessment body (CAB) | EN 60601-1:2006+A1:2013 (IEC 60601-1:2005+A1:2012) |
| Saudi Arabia | SFDA Approvals | - |
| Taiwan | Audit Report for Technical Cooperation Program | - |
| USA | cTUVus mark | ANSI/AAMI ES60601-1:2005+A1:2012 |
Medical Device Single Audit Program
Furthermore, the Medical Device Single Audit Program (MDSAP) offers you the possibility to fulfill the quality management requirements for an approval of your medical devices in the participating countries Australia, Brazil, Japan, Canada and the USA with only one audit.
For your medical electrical equipment, we also offer to carry out the CB procedure to facilitate international marketing. With a CB certificate and the CB test report, manufacturers can apply for the respective test marks of different target markets more quickly.
| Based on old directives | Based on new regulations | ||
|
Active and Non-Active Medical Devices (MDD)
The currently still valid EC Directive 93/42/EEC contains the requirements for active and non-active medical devices for the European market. |
European Medical Devices Regulation (MDR) 2017/745
As Notified Body for conformity procedures and certifications according to the MDR, we provide services throughout your transition to the new regulation and with the re-certification of your medical devices. | ||
|
In-Vitro Diagnostic Medical Devices (IVDD)
We are a Notified Body for the Directive 98/79/EC on in-vitro diagnostic medical devices and will perform the testing and certification of your quality management system and products. |
EU Regulation on In-Vitro Diagnostics (IVDR) 2017/746
As Notified Body for conformity assessment procedures and certifications according to IVDR, we provide services throughout your transition to the new regulation and the re-certification of your in-vitro diagnostic medical devices. |
Our Global Services
Our experts have extensive knowledge and many years of experience in the testing and certification of medical devices. With our Market Access Services , we support you in Europe and worldwide.
Our expertise
In the partnership with TÜV Rheinland, you benefit from 150 years of experience as a global, independent testing, inspection and certification organization. We also owe our international recognition to the active participation of our experts in international standardization committees. The knowledge gained from committee merges directly into our services for medical devices.
Our processes
Of course, flexibility, speed and transparency are important criteria for you during the certification process. As a reliable partner, we offer you direct contact with our experts and focus on personal and individual support for our customers.
Our laboratories
Through our global network of accredited laboratories, we are able to offer you consistent services worldwide. Our modern testing facilities are always state-of-the-art.
Join us on a tour of our medical device testing laboratory in Budapest!
Contact us and ensure the success of your medical product!
With the comprehensive conformity tests for medical devices and our QM system certifications, you prove the safety, quality and performance of your products.
Rely on the expertise of a renowned, international testing organization. We are able to offer all legally required tests worldwide and from a single source. This saves time and money and ensures the success of your product.
Achieve international market access now



_core_4_3.jpg)
_core_4_3.jpg)
