PSCI Audit Program

Enabling you to demonstrate responsible supply chain management and conformance with the PSCI principles
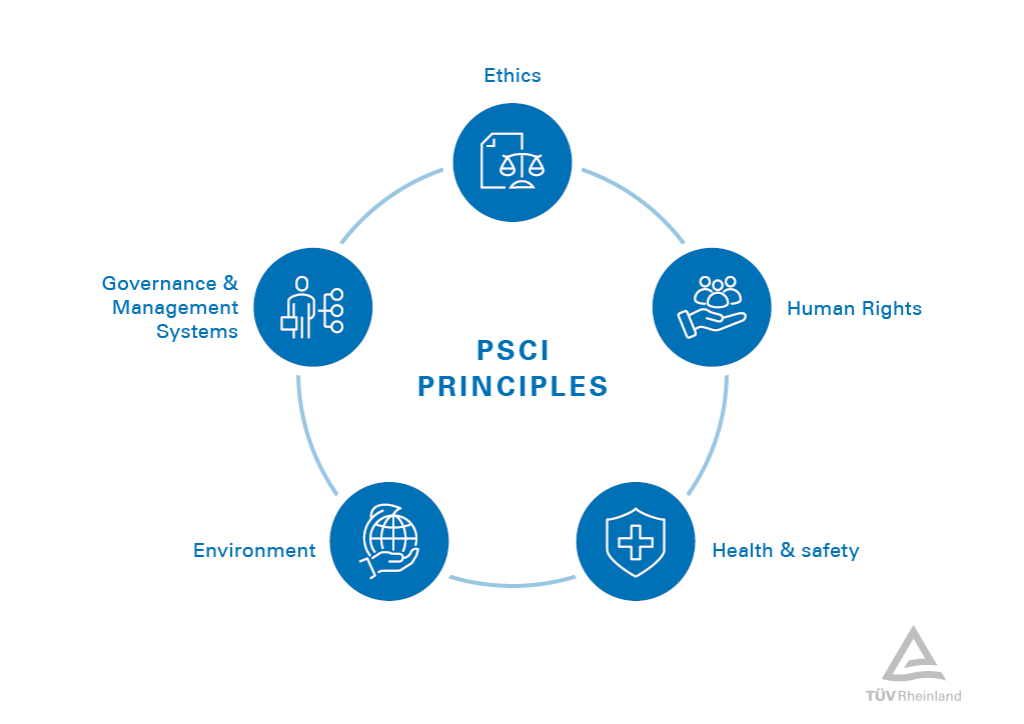
In the pharmaceutical and healthcare industry, the PSCI Principles for Responsible Supply Chain Management have become a widely recognized standard for defining industry expectations. These principles focus on five key areas: ethics, human rights, health & safety, environment, and governance & management systems. Demonstrating compliance with these principles can significantly enhance your company’s reputation and position within the sector.
As a globally approved audit service provider of the Pharmaceutical Supply Chain Initiative (PSCI), we bring extensive experience and expertise to the table. This enables us to effectively support companies across the pharmaceutical and healthcare industry - yours included - on their journey toward sustainable and responsible supply chain practices.
Get in touch with us to initiate the PSCI auditing process in your company.
Our PSCI audit program stands for socially responsible practices and provides the tools for meeting industry expectations
In an industry where ethics, human rights, environmental, health and safety concerns are defining the future, undergoing a PSCI audit is a clear signal to your business partners that your company prioritizes responsible practices and effective supply chain management. Our auditing services give you access to qualified, experienced audit teams around the world and ensure transparency in the auditing process. It’s a proven way to build trust among organizations active in the pharmaceutical and healthcare industry, and enables your company to communicate the values it stands for.
There are internal benefits as well. A PSCI audit pinpoints concrete risks along the supply chain and can identify and prioritize areas for improvement. It can lead to better awareness of both international & local regulations as well as industry expectations. By demonstrating that you meet the PSCI principles, you can qualify as a supplier to the industry, and members of the Pharmaceutical Supply Chain Initiative trust the PSCI supplier database to find reliable partners.
Our PSCI audit program
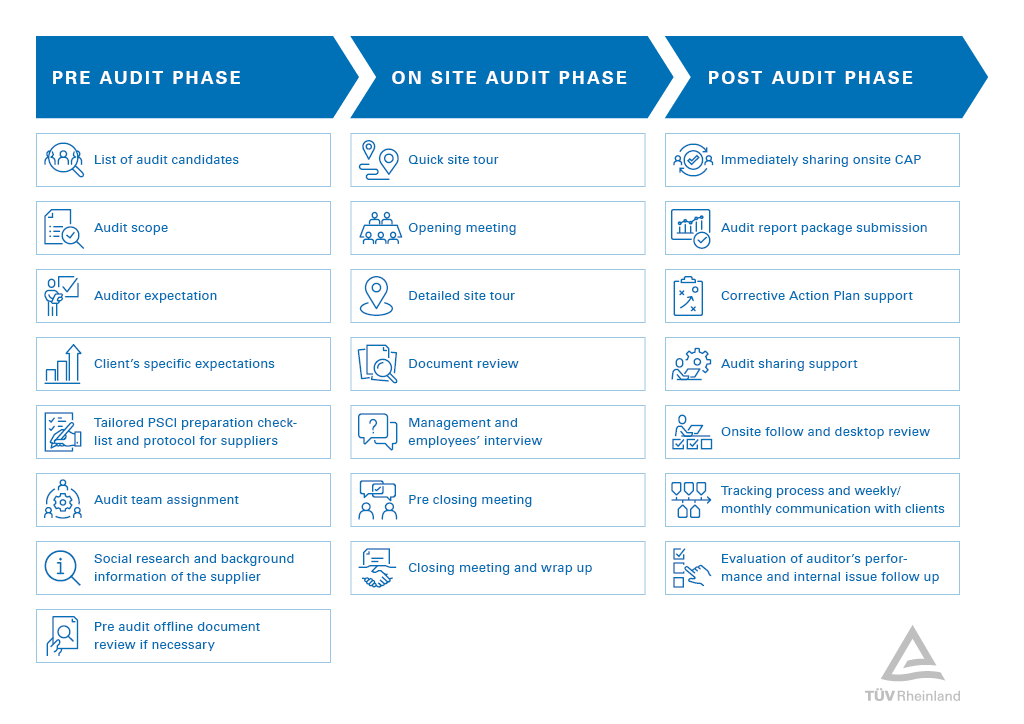
We conduct PSCI audits throughout the entire supply chain of international sourcing companies and for individual suppliers. Managed by a dedicated project team, we offer continuous and transparent reporting as well as central invoicing. To get started, contact us directly or reach a PSCI member company.
There is a 3-phase audit process, beginning with a pre-audit phase, continuing with the on-site audit and concluding with the post-audit phase. For large audit programs, status tracking is part of our transparent, holistic reporting process. It includes:
- Periodically updated tracking-lists for a clear overview and efficient control of the audit process.
- Periodic status-calls to ensure close communication between clients and our global project management.
As its global approved audit service provider, the PSCI trusts us and so can you
The Pharmaceutical Supply Chain Initiative recognizes our expertise in conducting social and ethical as well as health, safety, and environment (HSE) audits. We have already conducted several hundred audits in over 80 countries, and we continue to work closely with pharmaceutical & healthcare companies, suppliers and PSCI members to promote better ethics, human rights, health & safety, environment outcomes.
Our central project management and global project structure is a one-stop shop and guarantees high process quality at the same time. Our auditors are highly qualified and experienced in CSR, and HSE and undergo continuous professional development.
Contact us now. We look forward to showing you how a PSCI audit can benefit your company.
What you should know about the PSCI initiative and audit program
FAQs - PSCI Audit Program
| FAQs – Program Audit PSCI | 294 KB | Download |
Related services
Contact