Medical Device Training

Meet international market standards with our medical device training courses
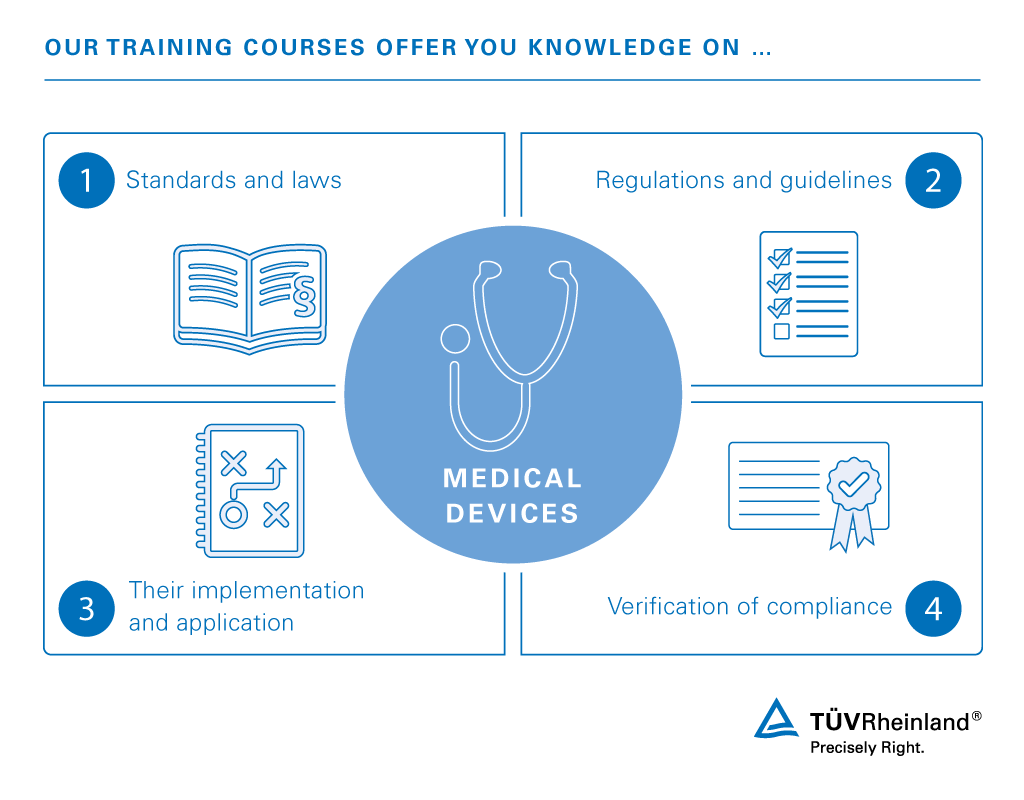
Companies producing medical devices and their professional staff comply with international standards, laws and regulations to demonstrate safety and quality. Industry professionals must be able to properly implement and apply global guidelines to appropriately verify product compliance. We provide training and certifications in diverse areas affecting the successful placement and competitive performance of medical devices in international markets.
Our expert instructors conduct practice-oriented seminars and courses for professionals working in the medical device and technology industry. We provide up-to-date specialist knowledge for strong and sustainable presence in European and international markets. Our training addresses crucial time-to-market reality as well as complex and diverse requirements for development, manufacturing, market introduction and monitoring.
We are a leading provider of professional trainings. Rely on our extensive training portfolio with expert instructors and recognized certifications.
Contact us to learn how to ensure your product is compliant to standards, regulations and laws relevant to your selected markets!
Compliant and competitive around the world with comprehensive medical device seminars
Our extensive portfolio of certification curriculums provides industry professionals the expertise to successfully access and succeed in challenging international markets. We make sure you fully understand the standards, regulations and laws applicable to your markets of choice. We provide flexible trainings allowing you to choose courses according to your individual needs. Our courses are designed to give you the tools you need to prove the safety and quality of your product. Our training programs for medical devices focus on providing you the practical knowledge to gain critical regulatory approvals. We show you how to meet regulatory standards to ensure the successful placement and competitive performance of your medical devices in tough global markets.
Comprehensive training for the medical device industry
Our comprehensive training for the medical device industry is presented in practical seminars and courses resulting in recognized certifications. Distinguished instructors provide detailed training for industry professionals such as:
- Managers and specialists
- Safety officers for medical devices
- Medical device advisors
- Regulatory affairs managers
- Regulatory compliance authorities
Modular training courses in the following areas enable you to make sure your products meet complex and diverse international standards, regulations and laws. Curriculums for TÜV certification or university degree equivalents for the medical device industry include:
- International approval procedures
Properly ensure the safety and quality necessary to gain approvals worldwide. - Sale of medical devices
Clearly communicate your commitment to regulatory compliance in every market. - In vitro diagnostics
Successfully design the development process, ensure good risk management and high usability throughout the product lifecycle. - Medical software
Legally implement medical software guidelines and standards. - Medical devices law
Dramatically reduce liability risk by implementing sound knowledge of the legal requirements. - Production hygiene and sterility
Correctly interpret standards requirements and put them into practice. - Product safety and serviceability
Confidently exclude hazards caused by medical devices and avoid misuse. - Process, quality and risk management
Reliably design processes, expeditiously identify defects and minimize risk.
Courses can be booked in any order and according to your individual needs.
Expert training and globally-recognized certifications
A leading provider of comprehensive trainings in all professional sectors, we engage interdisciplinary instructors sourced from our respected global network of experts. Our extensive portfolio of training programs is designed to ensure exceptional skill sets. The recognized certifications and degree equivalents set our graduates apart from the competition. We are your one-stop-shop for modern, relevant and flexible training.
Get in touch with us today to enhance your professional qualifications and improve your skills when dealing with product compliance and placement for international markets!
Our Sustainability Initiatives
Nothing less than the future is at stake. Companies, institutions, public authorities and each and every one of us can play a positive role in shaping the path to tomorrow. We provide you with comprehensive support to ensure that you operate safely, sustainably and efficiently for many years to come.
Contact